NSABP Members' Area
Password Protected - Access
Limited to NSABP Participating
Institutions Only
NSABP Foundation, Inc.
General NSABP Information
Financial Conflicts of
Interest Policy
Contact the NSABP
Employment

Clinical Trials Information
Clinical Trials Overview
Protocol Chart
Never Say Lost
Treatment Trials Information
Protocol B-51
Protocol B-52
Protocol B-53/S1207
Protocol B-55/BIG 6-13
Prevention Trials Information
Protocol P-1 - BCPT
Protocol P-2 - STAR
To report problems, ask
questions or make comments,
please send e-mail to:
Webmaster@nsabp.pitt.edu
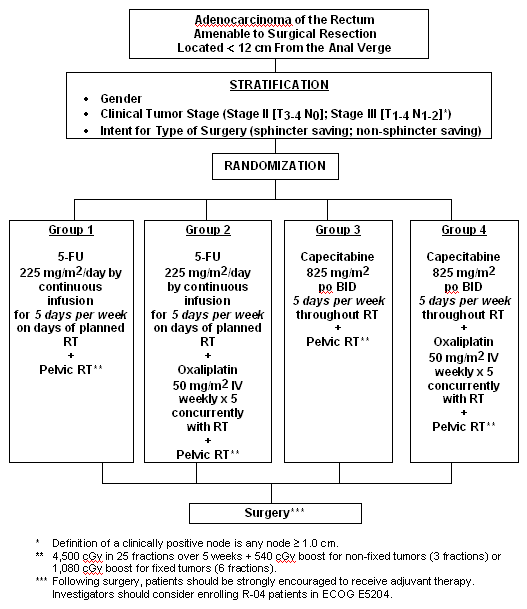
Protocol R-04
A Clinical Trial Comparing Preoperative Radiation Therapy and Capecitabine with or without Oxaliplatin with Preoperative Radiation Therapy and Continuous Intravenous Infusion of 5-Fluorouracil with or without Oxaliplatin in the Treatment of Patients with Operable Carcinoma of the Rectum
Specific Aims
Primary Aim
The primary aims of this study are to:
- Compare the rate of local-regional relapse in patients receiving preoperative oral capecitabine + radiotherapy (RT) to that in patients receiving preoperative continuous intravenous infusion (CVI) 5-FU + RT;
- Compare the rate of local-regional relapse in patients receiving preoperative oxaliplatin with those not receiving preoperative oxaliplatin.
The secondary aims of this study are to:
- Compare rate of cCR in patients receiving preoperative oral capecitabine + RT to that in patients receiving preoperative CVI 5-FU + RT;
- Compare rate of cCR in patients receiving preoperative oxaliplatin with those not receiving preoperative oxaliplatin;
- Compare rate of pCR in patients receiving preoperative oral capecitabine + RT to that in patients receiving preoperative CVI 5-FU + RT;
- Compare rate of pCR in patients receiving preoperative oxaliplatin with those not receiving preoperative oxaliplatin;
- Determine the increase in the number of patients able to undergo sphincter-saving surgery;
- Correlate genetic patterns and the presence or absence of specific tissue biomarkers with response and prognosis;
- Compare the impact of oral capecitabine versus CVI 5-FU on QOL in the setting of preoperative radiotherapy for rectal cancer;
- Evaluate the impact of oxaliplatin on neurotoxicity in the setting of preoperative radiotherapy for rectal cancer;
- Examine the differences in toxicity and convenience of care for the four chemotherapy treatment regimens;
- Describe the impact of the type of surgical management of rectal cancer on QOL at 1 year after surgical treatment.